Results 1 to 49 of 49
Thread: superheat
-
22-04-2007, 06:31 PM #1
superheat
hello guys i am new in this field of refrigeration . is there any formula to calcute asuperheat in low pressure. i know is to do with the expansion valve . do u need any special tools to mesure superheat . thank u guys.
-
22-04-2007, 07:20 PM #2
Re: superheat
Hi
http://www.refrigeration-engineer.co...ight=superheat
http://www.refrigeration-engineer.co...ght=superheati
There is lots more
Chheers BernardLast edited by bernard; 22-04-2007 at 07:22 PM.
-
23-04-2007, 07:34 AM #3
Re: superheat
Hi there,
Superheat is one of the basic definition you need in this field.
The simple explanation is this : When a liquid is boiling in a constant temperature (boiling point), say 5°C - and after a while all the liquid converts to gas and the gas temperature reaches to a higher temperature, say 12°C, then superheat is 12-5=7°C. In other word , the gas is heated 7°C more than the boiling temperature.
In the evaporator the same happens.
I suggest you study from basic principles until you can measure the superheat.
It is not difficult but you have to know the pressure-temperature relationship first.
Good luckEven Einstein Asked Questions
-
23-04-2007, 08:07 AM #4
Re: superheat
Hello,
you can use the HPT transmitter from Hansen Technologies. It gives a 4..20 mA Signal due to the superheat or subcooling, suitable for diferend kinds of refrgerants.
-
23-04-2007, 07:06 PM #5
Re: superheat
Information on the HPT sensor mentioned above from here...
http://www.hantech.com/default.asp
The site also contains some interesting data sheets and downloads.Brian - Newton Abbot, Devon, UK
Retired March 2015
-
28-04-2007, 04:22 PM #6
Re: superheat
thank you for effort lana.
i would to know how to work out superheat on the system .. let say system is running with r404 . suction pessure is about 3 bar. is there any calculation needed
-
28-04-2007, 04:48 PM #7
Re: superheat
Hi Marco,
To measure the superheat do the followings:
1- Take the suction pressure.
2- Go to refrigerant pressure and temperature chart and find the evaporating temperature (Te). Be careful about the pressure readings. The pressure you read from the gauge is 'gauge pressure". if your chart is absolute pressure then you have to convert the reading.
3- With a thermometer measure the suction line temperature neat the TEV bulb. (T2)
4- The superheat is T2-Te.
If you have Zeotropic refrigerant then there is a glide. For R404A the glide is 0.5°C so there isn't so much difference.
For other Zeotropic there are two temperatures for each pressure. You take the DEW POINT temperature as (Te).
Hope this helps.
CheersEven Einstein Asked Questions
-
29-04-2007, 11:51 PM #8
Re: superheat
Hi marco
Just want to add to lana's statement which is correct, i like to say it as you do it.
put your suction gauge on the suction service valve closest to the evapourator (make sure the gauges are the correct type for the refrigerant) look at what pressure it is and follow the gauge pin and write down the temperature shown on the gauge at that pressure. Use a thermosat and take a temperature reading of the suction pipe as it leaves the evapourator and write that down.
Pipe temperature minus gauge temperature equals super heat reading.
hope that helps
-
30-04-2007, 06:35 AM #9
Re: superheat
Hi Marco Blanco,
If you're interested, email me. I could forward you charts on method which is in pdf format.
-
30-04-2007, 06:45 AM #10
Re: superheat
Abdulazman, I'm interested too
 But for other reasons then Marco Blanco.
But for other reasons then Marco Blanco.
Do you have my email address?It's better to keep your mouth shut and give the impression that you're stupid than to open it and remove all doubt.
-
01-05-2007, 07:41 AM #11
Re: superheat
Hi Pete,

abdazman@****.comLast edited by frank; 01-05-2007 at 07:27 PM. Reason: Please use PM's for e-mail
-
05-05-2007, 03:05 PM #12
Re: superheat
thank you very much for help guys. lana.colments,abdulzaman, marco_1362@****.com
Last edited by frank; 07-05-2007 at 09:19 PM. Reason: Please use Private Messaging for e-mail addresses
-
08-05-2007, 03:23 AM #13
Re: superheat
Uh ohh is this a bad post? Sorry if it is just let me know I will remove...
You can measure superheat but do you know what it should be?
(Indoor wetbulb x 3) - 80 - outdoor ambient @ condenser)/2 = superheat needed +-5F on piston units.
Temp conversion from F to C or reverse:
degrees Celsius- = 0.56 (degrees Fahrenheit - 32)
- = (1.8 x degrees Celsius) + 32
-
08-05-2007, 04:32 AM #14
-
08-05-2007, 05:25 AM #15
Re: superheat
Your wetbulb gives you the idea of how much humidity is in the air....High humidity level air holds more heat...hidden heat (latent)
Specific heat-the amount of heat necessary to raise the temp of 1lb of a substance 1 degree F.
Specific heat of air...(.24/btu/F) (average)
Specific heat of water (1.00/btu/F)
So by knowing the wetbulb you now know how much hidden heat is in the air.... meaning how it will it affect the boiling point of the refrigerant.
By knowing the outside drybulb you will be able to find the condensing point of the refrigerant.
High efficiency is usually less than standard efficiency.
30F for standard efficiency....20-25F for high efficiency.....above ambient temps....But if you watch your subcooling you know when the ref is condensed all the way. Therefore you can find your condensing point.
The ref system is a cycle one affects the other...If you have lower outside temps you will condense the ref. much faster because it's a more efficient charge....greater the temp difference faster heat travels...If your condensing the liquid faster than design conditions it will starve the evaporator so you must be able to know all conditions to properly charge. If your at design conditions then you can just use your general superheating chart.....any other conditions and you must be able to account for them or else you'll over charge and can possibly cause damage to the system.
Make sense? If not I'll try a different way to explain it's kinda hard to explain because of all the other information you must know.
If you have no humidity in the air then you don't have to worry about wetbulb...but most places have humidity so you must factor it in....You must also wait 15 minutes before adding any charge to a system for equalization purposes.Last edited by BigJon3475; 08-05-2007 at 05:31 AM.
-
08-05-2007, 05:55 AM #16
Re: superheat
Hi there,
No problem so far.Your wetbulb gives you the idea of how much humidity is in the air....High humidity level air holds more heat...hidden heat (latent)
Specific heat-the amount of heat necessary to raise the temp of 1lb of a substance 1 degree F.
Specific heat of air...(.24/btu/F) (average)
Specific heat of water (1.00/btu/F)
So by knowing the wetbulb you now know how much hidden heat is in the air.... meaning how it will it affect the boiling point of the refrigerant.
OKBy knowing the outside drybulb you will be able to find the condensing point of the refrigerant.
High efficiency is usually less than standard efficiency.
30F for standard efficiency....20-25F for high efficiency.....above ambient temps....But if you watch your subcooling you know when the ref is condensed all the way. Therefore you can find your condensing point.
If the ambient temp is lower than design then the condensing temperature and therefore pressure will be lower.The ref system is a cycle one affects the other...If you have lower outside temps you will condense the ref. much faster because it's a more efficient charge....greater the temp difference faster heat travels...
If the ambient temp drops then the condensing pressure will drop too. If condensing pressure decreases too much then the TEV would not give design capacity and this is the reason of starved evaporator which leads to low suction pressure. NOT the "faster condensation".If your condensing the liquid faster than design conditions it will starve the evaporator so you must be able to know all conditions to properly charge.
Either at design condition or at different conditions, you need to measure super heat , sub cooling , LP, HP and ambient temp in order to charge the system properly.If your at design conditions then you can just use your general superheating chart.....any other conditions and you must be able to account for them or else you'll over charge and can possibly cause damage to the system.
I know.Make sense? If not I'll try a different way to explain it's kinda hard to explain because of all the other information you must know.
There is no absolute dry air. When the cooling system cools the air (if coil temp is lower than the air dew point) then you have de-humidification. When this happens, the air relative humidity increases.If you have no humidity in the air then you don't have to worry about wetbulb...
ONLY evaporative cooling process depends directly on Wet Bulb (constant wet bulb process). And this is the outside air wet bulb not the indoor.
I explained this already.but most places have humidity so you must factor it in....
Correct. But you have to wait for the system to equalize then measure the parameters. These parameters will dictate the amount of charge needed.You must also wait 15 minutes before adding any charge to a system for equalization purposes.
CheersEven Einstein Asked Questions
-
08-05-2007, 06:08 AM #17
Re: superheat
A TEV and a fixed bore are different devices.
We are talking about different systems here....
To charge with an expansion device you use subcooling...as the superheat is set."If the ambient temp drops then the condensing pressure will drop too. If condensing pressure decreases too much then the TEV would not give design capacity and this is the reason of starved evaporator which leads to low suction pressure. NOT the "faster condensation".
Correct....you asked how wetbulb affects superheat. If you are holding more heat in the air by having a higher relative humidity then it will affect super heat."Either at design condition or at different conditions, you need to measure super heat , sub cooling , LP, HP and ambient temp in order to charge the system properly."
The relative humidity decreases not increases if it increases you would be adding more humidity to the air. The indoor air is where you are trying to remove humidity from not the outdoor. The indoor is where the evaporator is hence where you need at least one superheat recording. You can take one right before the compressor to get a line set efficiency reading called total superheat but only if you take both at the evaporator leaving and the condenser going in. The evaporator is where the heat is that you need to remove it takes longer to remove heat with high humidity."There is no absolute dry air. When the cooling system cools the air (if coil temp is lower than the air dew point) then you have de-humidification. When this happens, the air relative humidity increases.
ONLY evaporative cooling process depends directly on Wet Bulb (constant wet bulb process). And this is the outside air wet bulb not the indoor."
A common misconception
Often the concept of air holding water vapor is used in the description of relative humidity. Relative humidity is wholly understood in terms of the physical properties of water alone and therefore is unrelated to this concept. Relative humidity is the ratio of the amount of water vapor in air to the maximum amount of water vapor that could be present if the vapor were at its saturation conditions.
By the time the equalize it still will not have removed all the humidity....so you must still take all readings...I think we are saying the same thing here."Correct. But you have to wait for the system to equalize then measure the parameters. These parameters will dictate the amount of charge needed."
And no offense I thought you were asking me because you did not know not because you were questioning if this was right or not.
Any superheat chart you ever look at has indoor wetbulb and drybulb and only outdoor drybulb.
I can't post a link because I don't have enough post yet sorry. Every manufacturer that post superheat charts with it's equipment requires wetbulb and drybulb indoor and outdoor drybulb only. You are conditioning the inside not the outside so taking the humidity from the inside is the only part that really matters. If you have subcooling on the condensing unit then you know the refrigerant is condensed and is losing heat beyond the condensing temp.
I do agree though you need all measurements to accurately add refrigerant.
Indoor Wetbulb
Indoor Drybulb
Outdoor Ambient
Low Pressure readings @ the same place you are taking temps for superheat.
High Pressure readings @ the same place you are taking temps for subcooling.Last edited by Peter_1; 08-05-2007 at 06:48 AM.
-
08-05-2007, 06:28 AM #18
Re: superheat
Hi there,
My statement is correct for TEV or cap tubes. If TEV is used then the superheat can be adjusted but with cap tube the super heat depends on the load and refrigerant charge.A TEV and a fixed bore are different devices.
We are talking about different systems here....
To charge with an expansion device you use subcooling...as the superheat is set
Correct....you asked how wetbulb affects superheat. If you are holding more heat in the air by having a higher relative humidity then it will affect super heat.
Completely wrongThe relative humidity decreases not increases if it increases you would be adding more humidity to the air. . Study Psychrometric my friend. Relative humidity is different than the actual humidity or moisture content.
. Study Psychrometric my friend. Relative humidity is different than the actual humidity or moisture content.
Correct.The indoor air is where you are trying to remove humidity from not the outdoor. The indoor is where the evaporator is hence where you need at least one superheat recording. You can take one right before the compressor to get a line set efficiency reading called total superheat but only if you take both at the evaporator leaving and the condenser going in.
OK.By the time the equalize it still will not have removed all the humidity....so you must still take all readings...I think we are saying the same thing here.
Let see what the others say, and believe me I know.And no offense I thought you were asking me because you did not know not because you were questioning if this was right or not.
CheersEven Einstein Asked Questions
-
08-05-2007, 06:31 AM #19
Re: superheat
What about the indoor dry bulb temperature? It can effect the targeted superheat up to 5 degrees fahrenheit. The formula given previously must assume a constant relative humidity factor since an indoor dry bulb temperature is not part of the formula.
-
08-05-2007, 06:37 AM #20
Re: superheat
I just explain this in different way.
The evaporator is inside the room and definitely the inside humidity influences the load on the coil. If the coil de-humidifies then a lot of energy is wasted to condense that moisture. When this happens then the superheat of course changes.
When designing the evaporator coil you can see how the air humidity effects the performance.
In order to check the refrigeration system, we have to measure the super heat, which is done with one thermometer and a suction gauge. Thermometer is used to take the evaporator outlet temp (pipe) and the LP is used to get the evaporating temperature. The different between these two temperatures is super heat.
CheersEven Einstein Asked Questions
-
08-05-2007, 06:46 AM #21
Re: superheat
Right I agree....for TEV you charge by subcooling not superheat. Although you want all your readings to look for things like restrictions or clogged evaps....ect..ect.."My statement is correct for TEV or cap tubes. If TEV is used then the superheat can be adjusted but with cap tube the super heat depends on the load and refrigerant charge."
"There is no absolute dry air. When the cooling system cools the air (if coil temp is lower than the air dew point) then you have de-humidification. When this happens, the air relative humidity increases.
ONLY evaporative cooling process depends directly on Wet Bulb (constant wet bulb process). And this is the outside air wet bulb not the indoor."
"Often the concept of air holding water vapor is used in the description of relative humidity. Relative humidity is wholly understood in terms of the physical properties of water alone and therefore is unrelated to this concept. Relative humidity is the ratio of the amount of water vapor in air to the maximum amount of water vapor that could be present if the vapor were at its saturation conditions.""Completely wrong . Study Psychrometric my friend. Relative humidity is different than the actual humidity or moisture content."
. Study Psychrometric my friend. Relative humidity is different than the actual humidity or moisture content."
Are you saying because the temp is going down the amount of moisture that the air can hold goes down also? Maybe I'm not following you. If the temp of the air decreases it holds less air...But if you decrease the temp of the air you have also removed more humidity to be able to decrease the temp. It will take longer to cool 80F air with 70% RH than it would take 80F with 50% RHLast edited by Peter_1; 09-05-2007 at 03:52 PM.
-
08-05-2007, 07:00 AM #22
Re: superheat
Measuring the subcooling if you have a TXV?
I don't have a SH chart because I don't need it. If your TXV was proper sized, then it's factory set at +/- 6K.
It tries to hold this 6K whatever pressure fluctuates and has nothing to do with the humidity. It's all about balancing pressures inside this TXV.
SH is theoretically only just till the suction valves, going in the condensor has nothing to do anymore with the SH.You can take one right before the compressor to get a line set efficiency reading called total superheat but only if you take both at the evaporator leaving and the condenser going in.
English is not my mother tongue but I think you add moistire to the air and not humidity.It's better to keep your mouth shut and give the impression that you're stupid than to open it and remove all doubt.
-
08-05-2007, 07:06 AM #23
Re: superheat
"Measuring the subcooling if you have a TXV?
I don't have a SH chart because I don't need it. If your TXV was proper sized, then it's factory set at +/- 6K.
It tries to hold this 6K whatever pressure fluctuates and has nothing to do with the humidity. It's all about balancing pressures inside this TXV."
TXV set superheat correct? You cant charge a system using superheat if it is set.....Therefore you must use subcooling which is not set by a TXV.
"A thermostatic expansion valve ( TXV) is precision device used to meter the flow of liquid refrigerant entering the evaporator at a rate that matches the amount of refrigerant being boiled off in the evaporator, This is it's main purpose but like all the other metering devices it also provides a pressure drop in the system, separating the high pressure side of the system from the low pressure side. thus allowing low pressure refrigerant to absorb heat onto it's self."
-
08-05-2007, 07:14 AM #24
Re: superheat
I see some mistakes in the formula as compared to a generic superheat chart. There is to much room for play with the +/- 5F....You guys showed me that much already..thank you.
Last edited by BigJon3475; 08-05-2007 at 07:19 AM.
-
08-05-2007, 07:32 AM #25
Re: superheat
I stand corrected or at least I think I do...LOL
As you move across the psychrometeric chart to cooler temps...as long as the humidity stays the same RH does increase. However you will have to remove that moisture before you can ever bring the inside temp down. So if you stay @ 40% RH and you go from 70F to 60F you drop approximately 15 grains of moisture per pound of dry air.
-
08-05-2007, 09:20 AM #26
Re: superheat
Hi,
What you explained is called "Percentage Saturation" NOT relative humidity.
Relative Humidity is the ratio of the partial pressure of water vapor in Air to the partial pressure of water vapor at saturation condition (at the same temperature).
You see : relative humidity is the ratio of two pressures.
There are three parameters defined for humidity.
1- Moisture content (kg/kg).
2- Percentage saturation (%).
3- Relative Humidity (%).
Moisture content is the actual humidity in the air. If physically moisture is added to the air this parameter will increase and vis versa.
Percentage Saturation is the ratio of the amount of water vapor in Air to the maximum amount of water vapor that could be present if the air were at its saturation conditions.
If air is cooled sensibly :
1- Moisture content stays constant.
2- Percentage Saturation increases.
3- Relative Humidity increases.
If air is cooled with de-humidification :
1- Moisture content decreases.
2- Percentage Saturation increases.
3- Relative Humidity increases.
Mathematically the value of relative humidity and percentage saturation are very close together. As temperature goes down this difference becomes less and if temperature goes up this difference increases.
@ 45°C RH=50% PS=47.48%
@ -15°C RH=50% PS=49.96%
Hope this helps
CheersLast edited by lana; 08-05-2007 at 09:23 AM.
Even Einstein Asked Questions
-
08-05-2007, 01:22 PM #27
Re: superheat
The amount of water vapor in the air at any given time is usually less than that required to saturate the air. The relative humidity is the percent of saturation humidity, generally calculated in relation to saturated vapor density.
RH=actual vapor density/sat. vapor density
The most common units for vapor density are gm/m3. For example, if the actual vapor density is 10 g/m3 at 20°C compared to the saturation vapor density at that temperature of 17.3 g/m3 , then the relative humidity is
RH=10g/m(cubed)/17.3g/(cubed)x100%=57.8%
Calculation
Careful! There are dangers and possible misconceptions in these common statements about relative humidity.Relative humidity is the amount of moisture in the air compared to what the air can "hold" at that temperature. When the air can't "hold" all the moisture, then it condenses as dew.
Straight out of the physics book.....
Maybe you call it something different over there....
-
08-05-2007, 03:20 PM #28
Re: superheat
The explanation you gave is the definition of "Percentage Saturation" not the relative humidity.
Relative humidity is the ratio of two partial pressures.
Check the ASHRAE or CIBSE of other psychometric books.
I don't call it something different here, you call it incorrectly over there.
Check this http://en.wikipedia.org/wiki/Relative_humidity
Specially " A common misconception " section.
Also check http://www.taftan.com/thermodynamics/PERCESAT.HTM
Thaaaaaaaank yooooooooouLast edited by lana; 08-05-2007 at 04:47 PM.
Even Einstein Asked Questions
-
08-05-2007, 06:00 PM #29
Re: superheat
That's where I got my information from the first time....
And you told me it was wrong.
"Relative humidity is the ratio of the amount of water vapor in air to the maximum amount of water vapor that could be present if the vapor were at its saturation conditions."
" Thaaaaaaaank yooooooooou"
No need to get arrogant how will that lead to anything being solved here or anyone learning? :;-)
"Relative humidity is the ratio of the amount of water vapor in air to the maximum amount of water vapor that could be present if the vapor were at its saturation conditions."
From wikipedia....
Since I can't post weblinks I am at a disadvantage here... sorry I will prove my point after 15 posts.
Here is a relative humidity calculator:
go to www johnsnhweather .com/rh.html
Add the l at the end of the link.....You can play around with different temps and different RH points...
So are you still saying the amount of moisture in the air inside the apartment does not affect superheat? Since that is what this thread is about...
-
08-05-2007, 06:12 PM #30
Re: superheat
Why would you not allow someone to post links? That doesn't really lead to a very fair discussion?
-
08-05-2007, 06:13 PM #31
Re: superheat
Hi there,
Go to the link and read.That's where I got my information from the first time....
And you told me it was wrong.
"Relative humidity is the ratio of the amount of water vapor in air to the maximum amount of water vapor that could be present if the vapor were at its saturation conditions."
Relative humidity is defined as the ratio of the partial pressure of water vapor in a gaseous mixture of air and water to the saturated vapor pressure of water at a given temperature. Relative humidity is expressed as a percentage and is calculated in the following manner:

where:
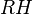 is the relative humidity of the gas mixture being considered;
is the relative humidity of the gas mixture being considered;  is the partial pressure of water vapor in the gas mixture; and
is the partial pressure of water vapor in the gas mixture; and  is the saturation vapor pressure of water at the temperature of the gas mixture.
is the saturation vapor pressure of water at the temperature of the gas mixture.
You see it is a ratio of two pressures.
Thanks.
No need to get arrogant how will that lead to anything being solved here or anyone learning? :;-)
This is a bad thing to say and I have nothing to say to you about this.
This is the definition of "Percentage Saturation". The Wikipedia definition is given above."Relative humidity is the ratio of the amount of water vapor in air to the maximum amount of water vapor that could be present if the vapor were at its saturation conditions."
From wikipedia....
I don't need software, I already have one and also I developed one.Here is a relative humidity calculator:
go to www johnsnhweather .com/rh.html
Add the l at the end of the link.....You can play around with different temps and different RH points...
As I said before (read my previouse post), moisture in the room adds to the load on the coil, so a lot of energy is used to condense this moisture. Definitely it effects the super heat (because it effects the load).So are you still saying the amount of moisture in the air inside the apartment does not affect superheat? Since that is what this thread is about
Good Luck with your study.
CheersLast edited by lana; 08-05-2007 at 06:18 PM.
Even Einstein Asked Questions
-
08-05-2007, 06:16 PM #32
Re: superheat
Specific humidity or moisture content of air is the ratio of the mass of water to the mass of dry air in a given volume of moist air.
Percentage saturation is defined as the ratio of the percentage saturation of air to the specific humidity of saturated air at the same temperature
Relative humidity is defined as the ratio of the partial pressure of water vapor in a gaseous mixture of air and water to the saturated vapor pressure of water at a given temperature. Relative humidity is expressed as a percentage and is calculated in the following manner.
Again we are saying the same thing. You feel your words are right and I'm wrong because why? We are on two different parts of the world? You just choose links from the exact same places I gave you the first time.
-
08-05-2007, 06:24 PM #33
Re: superheat
This is the correct definition of relative humidity. You hadn't given this definition till your last post.Relative humidity is defined as the ratio of the partial pressure of water vapor in a gaseous mixture of air and water to the saturated vapor pressure of water at a given temperature. Relative humidity is expressed as a percentage and is calculated in the following manner.
If you read the exact definition we wouldn't have this discussion, would we?Again we are saying the same thing. You feel your words are right and I'm wrong because why? We are on two different parts of the world? You just choose links from the exact same places I gave you the first time.
CheersEven Einstein Asked Questions
-
08-05-2007, 06:34 PM #34
Re: superheat
The wetbulb and I will repeat tells you the amount of moisture in the air when you compare it to dry bulb:
80F DB with a 80F WB will tell you that it has 100% RH
80F DB with a 77.67F WB will tell you that you have 90% RH
80F DB with a 73.4F WB tells you that you have 80% RH
80F DB with a 72.57F WB tell you that you have 70% RH
80F DB with a 69.98F WB tells you that you have 60% RH
80F DB with a 66.88 WB tells you that you have 50% RH
All of these measurements are taken at sea level or 29.92 inhg
If you cahnge the DB then the WB changes also:
70F DB with a 70F WB tells you that you have 100% RH
70F DB with a 67.96F WB tells you that you have 90% RH
70F DB with a 65.79F WB tells you that you have 80% RH
70F DB with a 63.48F WB tells you that you have 70% RH
Now lets look at different altitudes:
80F DB with a 77.59F WB tells you that you have 90% RH @ 27 inhg
80F DB with a 75.2F WB tells you that you have 80% RH @ 27 inhg
80F DB with a 77.72F WB tells you that you have 90% RH @ 25 inhg
80F DB with a 74.97F WB tells you that you have 80% RH @ 25 inhg
80F DB with a 77.5F WB tells you that you have 90% RH @ 23 inhg
80F DB with a 75.02F WB tells you that you have 80% RH @ 23 inhg
So forth and so on.....
-
08-05-2007, 06:40 PM #35
Re: superheat
This is absolutely correct as the air characteristic changes with the pressure.
We calculate or draw the psychometric chart for specific air pressure. The standard Psych. chart we use is for 1 bar. If you change the air pressure the "face" of the chart changes.
So you are right with your figures.
CheersEven Einstein Asked Questions
-
08-05-2007, 06:43 PM #36
Re: superheat
Okay well I agree that I did not give that definition although I did get my info from wiki but I had to edit because I cant copy and paste as you have done. I'm not allowed until 15 post.
I think we were saying the same thing either way. It's been a fun conversation but kinda redundant. I'm sure we will chat again.
I must return to work soon. Good day :-) I forced a lot of info in my brain thanks
-
08-05-2007, 06:51 PM #37
Re: superheat
No problem man, don't feel ....
I have been there, beleive me.
Basic definitions are the core to what we learn latter.
If you want to learn electricity then the first step is to learn WHAT IS THE ELECTRICITY? After that you go by the river.
I hope we both learn something from this discussion. It's been educational.
Good luck with whatever you do.
CheersEven Einstein Asked Questions
-
08-05-2007, 07:44 PM #38
Re: superheat
Hi BigJon
The restriction on posting links for newcomers to the forum is to stop single post advertising and spamming.
With your posting history I can't see it being too long before you are posting links.
If you wish to post a link to make your point in the meantime then just write it out using spaces instead of full stops and if it's a good link I'm sure one of the Mods will make it into a proper url.Last edited by frank; 08-05-2007 at 07:46 PM.
-
09-05-2007, 02:15 AM #39
Re: superheat
So I have a question then also....Is total superheat relevant? Meaning taking the superheat directly after the evaporator and also taking it at the service valves? Here is the conclusion I have come to and tell me if I'm wrong. If you take it directly after the evap. then you know the ref. is completely boiled off or at least in a sat. vapor state. If you take it right before entering into the compressor then you know the temp increase across the lineset.
But is that really needed?
Also if you have a long lineset and lets say the superheat on a fixed bore system is calling for 12DF by the time it gets to the condenser at the service ports its 30DF.
How do you know if it's cooling the compressor like it was designed to (besides if the winding thermostat is open)?
Is there a designed operating temp. for compressors?
Is there a time on a evaporator where the superheat leaving the evaporator will be 0 (either sat. liquid or sat. vap.) or should it always have some superheat say 1DF so you know it's at least a sat. vapor leaving the evap.
Thanks in advance for your help.
And since we were talking about psychrometrics I found something interesting upon some research.
A calculator for psychrometrics http://www.linric.com/webpsysi.htm and also http://www.handsdownsoftware.com/
I know those aren't for you guys but it could possibly help someone else out :-)Last edited by BigJon3475; 09-05-2007 at 02:36 AM.
-
09-05-2007, 04:29 AM #40
Re: superheat
Hi BigJon,
If I may, I would try to answer your questions.
You are right. he one taken at the evaporator outlet is called "useful super heat". The one taken at the compressor inlet is called "Total super heat" .So I have a question then also....Is total superheat relevant? Meaning taking the superheat directly after the evaporator and also taking it at the service valves? Here is the conclusion I have come to and tell me if I'm wrong. If you take it directly after the evap. then you know the ref. is completely boiled off or at least in a sat. vapor state. If you take it right before entering into the compressor then you know the temp increase across the lineset.
But is that really needed?
The useful super heat indicates the evaporation "efficiency", if you like, but the 'total super heat' indicates the suction line heat gain too.
The important one is the useful super heat which we measure it all the time. This doesn't mean that the 'total super heat' is not important. Usually, and almost always the suction line must be insulated, so you can see the picture here. If the line is not insulated and there is a lot of heat gain, the suction super heat would increase and this is the results :
1- High discharge temperature.
2- Increase compressor work.
3- Less cooling effect for the compressor.
In this question I lost you. What do you mean? The super heat at the compressor discharge is due to the compression precess.Also if you have a long lineset and lets say the superheat on a fixed bore system is calling for 12DF by the time it gets to the condenser at the service ports its 30DF.
How do you know if it's cooling the compressor like it was designed to (besides if the winding thermostat is open)?
Is there a designed operating temp. for compressors?
And yes, manufacturer states the best temperature at which the compressor can operate.
Yes, but if the super heat is zero then you have mixture of liquid and vapour going out.Is there a time on a evaporator where the superheat leaving the evaporator will be 0 (either sat. liquid or sat. VP.) or should it always have some superheat say DEF so you know it's at least a sat. vapor leaving the Eva.
We must always have between 5-8°C super heat to ensure there is no liquid going into the compressor.
With cap tube it is not in our hands, this is way an accumulator is recommended for such application.
Hope this helps
CheersEven Einstein Asked Questions
-
09-05-2007, 04:59 AM #41
Re: superheat
"In this question I lost you. What do you mean? The super heat at the compressor discharge is due to the compression precess.
And yes, manufacturer states the best temperature at which the compressor can operate."
Second part of your answer answered my question. I have not seen this in the literature but this would be available from manufacturer's websites possibly?
What I was trying to get at is we have been debating over the fact of having the correct superheat at the evaporator.....But then you might not be cooling the compressor off as it should be (depending on the length of the line set but only on really long linesets)
"Yes, but if the super heat is zero then you have mixture of liquid and vapour going out.
We must always have between 5-8°C super heat to ensure there is no liquid going into the compressor.
With cap tube it is not in our hands, this is way an accumulator is recommended for such application.
Hope this helps
Cheers"
All of my applications have an accumulator and I know how they work and I'm glad they are there :-).
Correct me if I'm wrong but isn't 5-8°C=41°F-46.4°F.. If that's correct I have been way off...
If that's correct I have been way off...
-
09-05-2007, 05:53 AM #42
Re: superheat
No No No.
Super heat is the difference between evaporator out let temperature and the evaporating temperature, NOT the actual line temperature.
Give you an example :
Evaporating temperature is -20°C (-4°F) and the evaporator outlet temperature is -15°C (5°F). The super heat is :
(-15°C-(-20°C))=5°C
or
(5°F-(-4°F))=9°F
In therms of °F normal super heat could be between 9°F and 15°F.
Hope this helps.Even Einstein Asked Questions
-
09-05-2007, 03:56 PM #43
Re: superheat
More then 20 years in business and eductaing new techs and still learning from the newly invented Log/P diagrams. Originally Posted by BigJon3475
Originally Posted by BigJon3475
Last edited by frank; 09-05-2007 at 08:02 PM.
It's better to keep your mouth shut and give the impression that you're stupid than to open it and remove all doubt.
-
09-05-2007, 05:12 PM #44
Re: superheat
Hi, BigJon3475

With ammonia you can go as low as 2K...or 3,6R, because for superheat we use absolute temperature scales Kelvin or Rankine but without sign ° for degree. Originally Posted by BigJon3475
Originally Posted by BigJon3475
Correct and not Originally Posted by BigJon3475
Originally Posted by BigJon3475

In terms of temperature level/value correct: 5°C=41°F, and 8°C=46.4°F,
but in terms of superheat wrong because:
due to different division on the scale we can say 1°C=1,8°F thus we have superheat of 5°C=9°F or 8°C=14.4°Fabove evaporating temperature meaning the difference and not a value
English is not my mother tongue but I hope this is kinda good explanation...
Best regards, Josip
It's impossible to make anything foolproof because fools are so ingenious...
Don't ever underestimate the power of stupid people when they are in large groups.
Please, don't teach me how to be stupid....
No job is as important as to jeopardize the safety of you or those that you work with.
-
09-05-2007, 05:26 PM #45
Re: superheat
I have to remember you guys are on a level I only hope to get on someday. Us in the service industry think of superheat as the temp above change of state.
If the boiling point of the refigerant is 40°F we think 44°F is 4°F of superheat.
This does help........me understand I don't know anything lol thanks.
-
09-05-2007, 05:28 PM #46
-
09-05-2007, 05:34 PM #47
Re: superheat
"With ammonia you can go as low as 2K...or 3,6R, because for superheat we use absolute temperature scales Kelvin or Rankine but without sign ° for degree."
I'm going to have to work on my conversions so it's easier for me to understand how the Engineers of the rest of the world describe the situations posed to them. I have scales and conversion charts we just don't use them much and I don't have much practice with them.
"Correct and not
In terms of temperature level/value correct: 5°C=41°F, and 8°C=46.4°F,
but in terms of superheat wrong because:
due to different division on the scale we can say 1°C=1,8°F thus we have superheat of 5°C=9°F or 8°C=14.4°Fabove evaporating temperature meaning the difference and not a value
English is not my mother tongue but I hope this is kinda good explanation...
Best regards, Josip "
"
Explained it well enough for me to understand so that's good....Thanks guys for taking the time to explain this to this "newbie"..... Refrigeration fascinates me and I hope my situation will lead me more into the engineering side of it instead of being a tech.Last edited by Peter_1; 10-05-2007 at 07:10 AM.
-
09-05-2007, 05:54 PM #48
Re: superheat
Hi, BigJon3475

Everything is ok, just stay with us and take some time for surfing on old posts and I believe you can learn a lot of things
Whenever you are in doubt come back with your question (pls not too much)
(pls not too much)
Also..
term boiling point we use only for refrigerants at atmospheric pressure for other pressures refrigerant boiling point is saturation temperature. Originally Posted by BigJon3474
Originally Posted by BigJon3474
Best regards, Josip
Last edited by Josip; 09-05-2007 at 06:34 PM. Reason: add some text
It's impossible to make anything foolproof because fools are so ingenious...
Don't ever underestimate the power of stupid people when they are in large groups.
Please, don't teach me how to be stupid....
No job is as important as to jeopardize the safety of you or those that you work with.
-
09-05-2007, 06:13 PM #49
Re: superheat
Hi peter_1

Did you receive my email?
Was it helpful?
Hope its helps in your quest.
I wish to thank the mod_r who corrected my post on the email address.
Adios Amigo






